 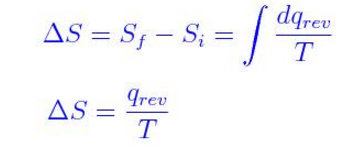
Increasing distance in alkaline-earth-metal/carbon #ns"/"2s# orbital ground-state energies (increasing the number of nonbonding = "52 = "65.7 = "93 J/mol"cdot"K"#Īnd for something more complex like buckminsterfullerene, the entropy is significantly higher because it can assume more microstates for a given macrostate (more molecular motions possible, more vibrational modes, etc).\): A Generalized Plot of Entropy versus Temperature for a Single Substance. H U+ (PV) qrev + wrev + (P V) where: qrev and wrev are the most efficient (reversible) heat flow and work, respectively. Generally a greater difference in radii between cation/anion corresponds to a greater internuclear distance and thus a weaker bond.Īnd since I recall carbonates decomposing at higher temperatures (which I found weird when I first learned it), it doesn't hurt to check alkaline earth metal carbonates going down the periodic table (wikipedia). Starting from the first law of thermodynamics and the relationship of enthalpy H to internal energy U: U qrev +wrev. Naturally the increase in charge magnitudes correlates with an increase in bond order or bond strength. The stronger the bond, the lower the magnitude of the entropy because the lower the number of microstates available to the solid. #Omega# is the number of microstates consistent with a chosen macrostate, which is proportional to the number of observable "snapshots" of molecular motion.One of the most popular thermodynamics equations is: With solids, I would not expect pressure to significantly alter any entropy patterns they already have for small values for pressure that would otherwise be significant for gases.įor di/polyatomicsolids, we can consider either the complexity or the bond strength, as it relates to the number of "ways" it can exist. On the other hand, the change in volume of a liquid is appreciably low upon small increases in pressure that should substantially compress a gas, so the change in pressure of a liquid makes a smaller negative contribution to the change in entropy. Assuming ideal behavior, calculate the entropy of 3. Assuming ideal behavior, calculate the entropy of 3.00 moles of He(g) at 25 C and a volume of 3000.0 L. DISCUSSION Gibbs Paradox illustrates an interesting aspect of entropy. If you look up the enthalpy of fusion for ice in a table, you would get a molar enthalpy of 6.01 kJ/mol. (Regardless, the entropy of the universe is #>= 0#.) The molar entropy of helium gas at 25 C and 1.00 atm is 126.1 J K-1 mol-1. Calculate entropy change when 36.0 g of ice melts at 273 K and 1 atm. Therefore, if pressure increases, a negative contribution is made on the change in entropy of an ideal gas, but depending on the change in temperature, the actual change in entropy for the system might be positive or negative. Since #C_P# for a monatomic ideal gas is #C_V + nR = 3/2nR + nR = 5/2nR#, with #C_V# as the constant-volume heat capacity and the #3/2# coming from the three linear degrees of freedom ( #x,y,z#), this becomes: When we relate pressure then, to entropy, with #S = S(T,P)#:įor an ideal monatomic gas, #PV = nRT#, so: 
Which is what you would get for the Maxwell relation. Thus, utilizing this relationship and invoking the Product Rule on #d(PV)#, we get: #w_"rev" = -PdV#.Īnother relationship that relates with heat flow is the one for entropy and reversible heat flow: #q_"rev"# and #w_"rev"# are the most efficient (reversible) heat flow and work, respectively.#\mathbf(DeltaH = DeltaU + Delta(PV)) = q_"rev" + w_"rev" + Delta(PV)# Starting from the first law of thermodynamics and the relationship of enthalpy #H# to internal energy #U#:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
